|
June 2001
Diagnosis: Herpes zoster
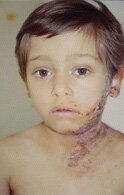 Varicella-Zoster is the virus that causes chickenpox and herpes zoster (shingles). Infection with this virus can be serious in a person with HIV/AIDS. Following primary varicella-zoster infection, the virus remains in the body in the latent form, and can be reactivated multiple times in individuals with immune suppression. The rash that appears when the virus is reactivated is called herpes zoster. A prodrome of burning pain and pruritis commonly precedes the appearance of a classic erythematous, vesicular rash. During the course of healing the rash becomes crusted and gradually disappears. Varicella-Zoster is the virus that causes chickenpox and herpes zoster (shingles). Infection with this virus can be serious in a person with HIV/AIDS. Following primary varicella-zoster infection, the virus remains in the body in the latent form, and can be reactivated multiple times in individuals with immune suppression. The rash that appears when the virus is reactivated is called herpes zoster. A prodrome of burning pain and pruritis commonly precedes the appearance of a classic erythematous, vesicular rash. During the course of healing the rash becomes crusted and gradually disappears.
Acyclovir, an antiviral medication, can be given either orally or intravenously. The pediatric oral dose is 20 mg/kg every 6 hours for 5 days. The adult dose is 800 mg four times a day for five to seven days. Acyclovir can cause pancytopenia, particularly when given in conjunction with zidovudine. It is important to increase the intake of oral fluids while receiving acyclovir to avoid crystalluria and possible acute renal failure.
Adjuvant pain medications may be required for pain control. Acetaminophen 10-15 mg/kg every 4 to 6 hours as needed (maximum 2.6 gm/day) can be used for children, and 650 mg every 4 to 6 hours (maximum 4 gm/day) can be given to adults. If the pain is not relieved, acetaminophen with codeine can be used. The pediatric oral dose, based on the codeine content, is 0.5-1 mg/kg every 4 to 6 hours. Adults can take 1 to 2 tablets (acetaminophen 300 mg and codeine phosphate 30 mg) every 4 hours (maximum 12 tablets/day).
Pruritis can be managed with oral diphenhydramine hydrochloride. The pediatric dose is 1 mg/kg every 6 hours as needed. Adults may take 25-50 mg every 4 to 6 hours as needed. Frequent baths in a tub with lukewarm water and colloidal oatmeal also may provide topical relief. Fingernails should be kept trimmed to a short length. It may be necessary to encourage the use of soft cotton gloves with children who insist on scratching the affected area.
There is a vaccine available to protect patients against initial infection with varicella-zoster virus. In May 1999, the U. S. Centers for Disease Control and Prevention updated the varicella vaccine recommendations for HIV-infected children. The new recommendations state that if an HIV-infected child has a CD4+ lymphocyte percentage greater than 25, the vaccine may be administered. Herpes zoster does not appear to occur any more commonly among vaccine recipients than among those with a history of natural varicella.
|